Myeloma Cancer: New Dynamic Risk Tool and CAR T Shift Force Earlier Action
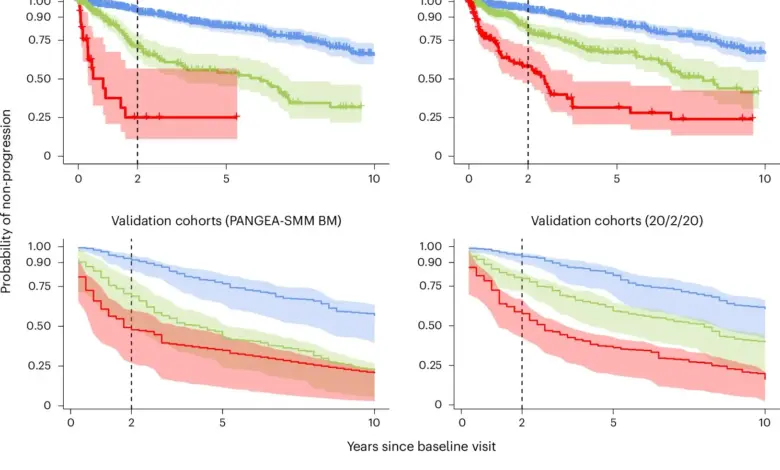
Investigators and clinicians have unveiled a dynamic risk model and changing treatment patterns aimed at earlier interception of myeloma cancer; the model and practice shifts were described in recent scientific and market reports. The PANGEA-SMM model was trained and validated on a cohort assembled from seven international centers and identifies four evolving biomarkers that signal imminent progression. Market analytics data dated March 24, 2026 (ET) show clinicians are increasingly planning to use CAR T-cell therapies earlier in the disease course, reshaping frontline strategy.
Enhanced dynamic risk stratification
Researchers developed the Precursor Asymptomatic Neoplasms by Group Effort Analysis SMM risk models (PANGEA-SMM) using longitudinal clinical and biological data from 2, 344 patients to predict progression from smoldering multiple myeloma to active disease. Four evolving biomarkers were significantly associated with shorter time to progression: an M-protein increase ≥0. 2 g dl−1, an involved/uninvolved serum free light chain ratio increase ≥20, a creatinine increase >25%, and a hemoglobin decrease ≥1. 5 g dl−1. The PANGEA-SMM models outperformed established static models, including the 20/2/20 and IMWG models, yielding a C-statistic of 0. 79 for predicting progression; performance remained high even without prior biomarker history or a recent bone marrow biopsy (C-statistic = 0. 78 in those scenarios).
Myeloma Cancer: Open-access PANGEA-SMM tool could improve monitoring
The team presents PANGEA-SMM as an easy-to-use, open-access tool that dynamically tracks routine lab measures already collected in follow-up, enabling clinicians to monitor trajectories rather than single snapshots. Dana-Farber Cancer Institute investigators and co-senior authors emphasized the model’s clinical accessibility: Dr. Irene Ghobrial, Director of the Center for Early Detection and Interception of Blood Cancers at the Dana-Farber Cancer Institute, said, “By watching the speed and direction of the disease’s trajectory, the tool can more accurately identify patients at high risk who need early treatment, while sparing those with stable disease from unnecessary interventions. ” Co-senior author Lorenzo Trippa, a computational biologist, is named among the model developers who validated PANGEA-SMM against established scores and made validation tools available for comparison.
CAR T-cell therapy moving to frontline as myeloma treatment
Market analysis from Spherix Global Insights indicates that clinicians treating multiple myeloma anticipate CAR T-cell therapies becoming standard first-line options. The report notes that recently approved CAR T products designed to bind BCMA use autologous T-cells collected from patients, engineered in the laboratory, and returned to deliver potent anti-myeloma effects. Spherix Global Insights said, “Taken together, the Q1 2026 findings suggest that the [multiple myeloma] treatment paradigm is entering a new phase: one defined not just by expanding options, but by a willingness to rethink long-standing sequencing conventions. ” The company also noted logistical barriers are becoming less of a hurdle as clinicians gain familiarity, and it projected increases in the proportion of patients receiving CAR T as second-line and third-line therapy over the near term.
Quick context: smoldering multiple myeloma is a biologically heterogeneous precursor state with highly variable time to progression; static scores like 20/2/20 and IMWG rely on single-time biomarkers and bone marrow biopsy metrics that can miss evolving risk. The Food and Drug Administration has recently approved a therapy for high-risk smoldering disease, underscoring clinical momentum toward early intervention for patients at imminent risk.
What’s next: clinicians and centers will test PANGEA-SMM in practice while monitoring how rapidly CAR T-cell therapies move earlier in treatment sequencing; watch for further validation work, broader clinical uptake of the open-access tool, and real-world data on how dynamic monitoring changes referral and treatment timing. Stakeholders will track these developments closely to determine whether earlier identification and earlier use of potent therapies together reduce progression and preserve organ function in patients with myeloma cancer.





